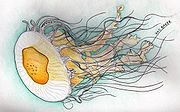
Phacellophora camtschatica
From marinelife1011
Phacellophora camtschatica is a member of the phylum Cnidaria and is commonly referred to as the fried-egg or egg-yolk jellyfish. It is called this because of its yolk-like bell that is surrounded by milky-transclucent edges. A warm-water species,Cotylorhiza tuberculata, typically found in the Mediterranean Sea, Aegean Sea, and Adriatic Sea, is also popularly called a fried egg jellyfish. This species is much different looking with many bright colors and would not be easily confused with P. camtschatica in situ.
| Fried Egg Jellyfish | |
|---|---|
 |
|
| P. camtschatica artwork by Rick & Cory Baker | |
| Scientific classification | |
| Kingdom: | Animalia |
| Phylum: | Cnidaria |
| Class: | Scyphozoa |
| Order: | Semaeostomeae |
| Family: | Ulmaridae |
| Genus: | Phacellophora |
| Species: | camtschatica |
| Binomial name | |
| Phacellophora camtschatica Brant, 1835 |
|
Contents |
Description and Phylogeny
Morphology
In all jellyfish species the exumbrella is the top-side of the bell and the subumbrella is the underside. The bell of P. camtschatica can measure up to 60 cm in diameter or 2 feet across. The central yellow mass is gonad tissue surrounded by a transparent or whitish margin. The bell margin generally consists of 16 clusters of tentacles (25 per cluster); 4 perradial, 4 interradial, and 8 adradial subumbrellar as well as 16 lappets (flaps) and 16 rhopalia (sensory structures).[1] The tentacles are sticky with only a mild sting and can extend 10-20 feet in length. Tentacle length is directly correlated with bell diameter, with the maximum length of tentacles approximately 17 times the bell diameter.[2] The oral arms are massively folded, but relatively short. [3]
Evolution
Within the phylum Cnidaria; Medusozoa, Hydrozoa and Cubozoa appear to be strongly monophyletic. In contrast, the taxa classified as Scyphozoa may not form a clade. Instead, scyphozoans appear to be paraphyletic with respect to Cubozoa. Phylogenetic results indicate that the polyp preceded the medusa in terms of evolution.[4]
Reproduction and Behavior
Reproduction
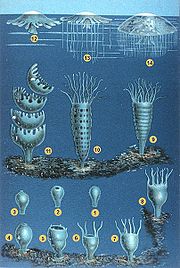
This jellyfish, like all Cnidarians, alternates between two life stages; polyp and medusa. Male and females reproduce sexually. Males release actively swimming sperm into the water column. Some of this sperm then enters the mouth of the female. Once fertilization has occured the free swimming ciliated planulae (larvae) then leave the mother and enter the water column. After several days of swimming the planulae settle and attach to a hard substrate on the sea floor. .[5] Once sessile the planulae develop into scyphistomae (polyps). P. camtschatica polyps undergo 2-, 4-, 8-,and 16-tentacled stages of development. The polyps reproduce asexually by transverse fission at thier oral ends. This metamorphosis is called strobilization or budding. Strobilae produce 8-15 juvenile medusae known as ephyrae during each cycle of replication. Once liberated into the water column the ephyrae then grow into sexually mature medusae in approximately 9 months.[1]
Lifespan
There is no exact information regarding the lifespan of P. camtschatica. Since the medusae are high in abundance only during the warmer months of the year it is reasonable to say that they most likely expire shortly after becoming sexually mature. The knowledge regarding polyps is even more limited, but they are most likely perennial, far outliving the medusae they produce.[1]
Foraging Behavior
P. camtschatica do not from large blooms, but they can be abundant.[6] Since they are most often found in locations where there is a high abundance of prey it has been suggested that they are either capable of finding their prey, or that they move less after encountering their prey. P. camtschatica uses two strategies for efficent prey capture: ambush and crusing. When they are motionless or slowly pulsating while drifting with extended tentacles over a large area they are termed 'ambush predators.' This strategy is more appropriate for rapidly moving species who get caught up in the jelly's tentacles. When they are swimming vertically in search for slow moving prey they are 'cruising predators.' Vertical movement and vertical tentacle deployment maximizes the probability of encountering prey which swim horizontally. When they ascend the water column they often are seen slowly spiraling which causes their tentacles to spread out over large areas. [2]
Symbionts
Several planktonic Cnidarians form symbiotic relationships with other marine invertebrates and vertebrates. P. camtschatica is host to the amphipod species Hyperia medusarum as well as the graceful crab, Cancer gracilis during its larval and juvenile stages. Megalopae (second larval stage) and juveniles of C. gracilis congregate on the bell and oral arms, but do not burrow into the bell or gonads of its host. They do however ingest its tentacles. During its naupliar stagesC. gracilis steals food from its host, but as it gets older it potentially benefits the fried-egg jelly by consuming H. medusarum, who are parasites to P. camtschatica. When feeding on its host H. medusarum feasts almost exclusively on tentacles.[7]
Ecology and Distribution
Geographic Range
The P. camtschatica occurs worldwide in eastern Pacific temperate waters from the Gulf of Alaska to Chile, in the Atlantic, and the Mediterranean. .[8]
Habitat
This species is pelagic. They prefer open temperate oceans, but are also seen near shores. Since ephyrae are released from benthic polyps it is likely that there is high polyp proliferation near shores where there are more hard substrates.[6]
Trophic Strategy
P. camtschatica feed on gelatinous zooplankton and other medusae. It is a voracious predator of its Cnidarian relative the moon jelly (Aurelia sp.). They appear to be top down predators and prefer food that they can easily manipulate.[1]This species and other gelatinous zoooplankton can exert significant pressure, to the point of initiating trophic cascades. When their populations are prolific (most likely from anthropogenic causes) there are significant changes in the structural food web and predator competition.[6]
Conservation
Conservation Status
P. camtschatica has not been evaluated by the International Union for Conservation of Nature (IUCN), but seems to be prevalent within its range and is not considered threatened.[9] There are however many other marine animals that rely on this and similar species for food including sea turtles, marine birds and at least 50 species of fish. These predators often mistake discarded plastic bags for jellies and get sick or die from consumption.[8]
Relevance
Since medusae of P. camtschatica prey upon gelatinous zooplankton and other medusae it is possible that they could aid in limiting populations of other pestiferous species of jellyfish. If this species were to become extinct or threatened it is likely that there would be a dramatic increase in the overall abundance of their prey.
Correlations between increases in jellyfish abundance and overfishing/eutrophication have already been established.[10][1][6][11][12] Because they respond to changes in their local ecosystems P. camtschatica and other jellyfish could act as indicator species.[1]
References
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 WIDMER, C.L. 2006. Life cycle of Phacellophora camtschatics (Cnidaria: Scyphozoa). Invertebrate Biology 125(2): 83-90.
- ↑ 2.0 2.1 STRAND, S.W., HAMMER, W.M. 1998. Predatory behavior of Phacellophora camtschatica and size-selective predation upon Aurelia aurita (Scyphozoa:Cnidaria) in Saanich Inlet, British Colubia. Marine Biology. 99: 409-414.
- ↑ http://www.wallawalla.edu/academics/departments/biology/rosario/inverts/Cnidaria/Class-Scyphozoa/Order-Semaeostomeae/Family-Ulmaridae/Phacellophora_camtschatica.html
- ↑ COLLINS, A.G. 2002. Phylogeny of Medusozoa and the evolution of cnidarian life cycles. J . EVOL. BIOL.15: 418-432.
- ↑ http://www.dnr.sc.gov/marine/pub/seascience/jellyfi.html
- ↑ 6.0 6.1 6.2 6.3 SUCHMAN, C.L., BRODEUR, R.D. 2005. Abundance and distribution of large medusae in surface waters in northern California Current. Deep-Sea Research II.52:51-72.
- ↑ TOWANDA, T., THUESEN, E.V. 2006. Ectosymbiotic behavior of Cancer gracilis and its trophic relationships with is host Phacellophora camtschatica and the parasitoid Hyperia medusarum. MAR. ECOL. PROG. SER. 315: 221-236.
- ↑ 8.0 8.1 http://www.montereybayaquarium.org/animals/AnimalDetails.aspx?enc=n3f4wmcSJaM7gUir8hqTxQ==
- ↑ http://www.eol.org/pages/203499
- ↑ MILLS, C.E. 2001. Jellyfish blooms: are populations increasing globally in response to changing ocean conditions?. Hydrobiologia 451: 55-68.
- ↑ THUESEN, E.V., RUTHERFORD JR, L.D., BROMMER, P.L., GARRISON, K., GUTOWSKA, M.A., TOWANDA, T. 2005. Intragel oxygen promotes hypoxia tolerance of scyphomedusae. J. Experimental Bio. 208: 2475-2482.
- ↑ http://pugetsoundscienceupdate.com/pmwiki.php?n=Chapter2a.Jellyfish
External Links
A video of Phacellophora camtschatica swimming in the water column
Photos
Pic 1. Baker, R. &. C. 2011
Pic 2. Sacco, V & D. http://wapedia.mobi/en/Jellyfish. 2/20/2011.
Pic 3. Baker, C. 2010.
Pic 4. World News. http://article.wn.com/view/2010/04/23/Giant_deep_sea_jellyfish_filmed_in_Gulf_of_Mexico/ (screen shot). 02/21/2011.
Pic 5. Baker, C. 2011.